
The Food Sensitivity Controversy
August 23, 2018
-
Indigestion, IBS, Reflux
-
Anxiety, Depression, Sleep Difficulty
-
Eczema and Rashes
-
Fatigue, Joint and Muscle pain/stiffness
-
Gluten Sensitivity
We Now Have Answers
Food sensitivities have been a controversial topic within medicine. The opinions range from the idea that food sensitivities do not exist, to they are a significant potential cause of many health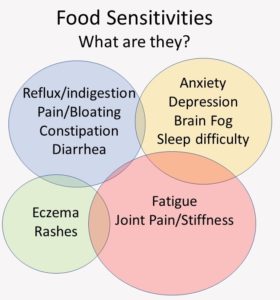 problems. As is often the case, much of this controversy persists because of a gap in the levels between current evidence and clinical practice. The best evidence examining this area has come from studying the most common food sensitivity, gluten sensitivity.
problems. As is often the case, much of this controversy persists because of a gap in the levels between current evidence and clinical practice. The best evidence examining this area has come from studying the most common food sensitivity, gluten sensitivity.
There are 3 classes of allergic immune reactions. Type I is called true allergy and is hallmarked by fairly rapid onset of symptoms driven by IgE antibodies. They can also be demonstrated with skin testing. Types II and III are called sensitivities and are typically delayed or gradual onset making them difficult to associate with their specific triggers.
Type II sensitivities are associated with IgA, IgG and IgM antibodies. Type II sensitivities may also cause “cross reactivity” where self tissue becomes confused with the food component triggering autoimmunity or making antibodies against one’s own tissue. Celiac disease, the most noted of these reactions, is associated with positive blood tests demonstrating IgG antibodies against gluten and also an enzyme in the small intestinal tissue. These “self antibodies” that develop by cross reactivity to gluten peptides begin to destroy the small intestinal lining.
More recent studies have suggested that the minority of persons with gluten sensitivity will go on to produce cross reactive antibodies resulting in celiac disease. Gluten sensitivity without celiac disease has been termed non-celiac gluten sensitivity or NCGS. NCGS is estimated to be 8-12 times more common than celiac disease. It is also highly symptom producing but without the destruction of the small intestinal lining as occurs in celiac disease.
Symptoms of NCGS tend to cluster in 4 systems:
Digestive tract
-
Indigestion
-
IBS
-
Reflux
Muscles and Joints
-
Stiffness
-
Pain
-
Swelling
Nervous System
-
Brain fog
-
Anxiety
-
Sleep dysfunction
Skin
-
Eczema
-
Rashes
-
Acne
Other Manifestations
-
Chronic Fatigue
-
Fibromyalgia
NCGS has been hotly debated in medicine. The primary reason has been the lack of a consistent objective test with which to “demonstrate” the disorder with a biomarker. Biomarkers are something that can be objectively measured such as an antibody in the blood which is present in those with a disorder and absent in those without. The difficulty with this in gluten sensitivity is that gluten antibodies are only present in about half of the subjects with symptoms thought to be related to gluten and which resolve when gluten is withheld. The reason for this is that type of NCGS symptoms can result from two different parts of the immune system. While type II sensitivities will produce gluten antibodies, type III sensitivities do not. They are produced by a branch of the immune system called the innate immune system.
The innate immune system is the first to respond to immune challenge by inducing primarily inflammation. It also attempts to trigger a response from the acquired immune system that produces antibodies. For about half of subjects with NCGS this second step does not occur, and they do not produce antibodies or a measurable biomarker. They simply have an ongoing inflammatory reaction trigger their symptoms.
A new study has added clarity to the divide between the “no antibodies, no problem” supporters and “inflammatory symptom” supporters. The study looked at markers which indicate damage to the small intestinal lining which is associated with “leaky gut”. Bacterial molecules are too large to be absorbed through the small intestinal wall. Inflammation of this barrier, however, causes the pores to become enlarged letting bacterial components enter the body and trigger inflammation in other areas.
The study compared normal subjects, patients with celiac disease and patients with NCGS. The protein marker of leaky gut was elevated only in the NCGS subjects. Similarly, 2 markers of bacterial components entering the system were also elevated only in NCGS subjects. The researchers concluded that there is a state of immune inflammatory activation caused by leakage of the intestinal barrier only in the NCGS subjects.
The bottom line is that there is now an accurate biomarker that demonstrates a food sensitivity. As Alessio Fasano, MD, Chair of Pediatric Gastroenterology at Massachusetts General Hospital and an authority on gluten sensitivity starts many of his lectures with “The gut is not Las Vegas”. He is referring to the fact that while the inflammation associated with gluten sensitivity begins in the gut, inflammation does not stay in the gut; it travels throughout the body and may cause any of the symptoms discussed above. Now we know how.
Uhde et al. Intestinal cell damage and systemic immune activation in individuals reporting sensitivity to wheat in the absence of coeliac disease. Gut, 2016;65:1930–1937.