
Turning the Lights Back on in Alzheimer’s Disease
July 20, 2021
One of the early findings in Alzheimer’s disease is that neurons or brain cells struggle to produce enough energy to function and thus begin to die off. One of the definitive tests to help diagnose early disease is an FDG PET scan. This test involves injecting a radiotracer glucose before obtaining a PET scan.
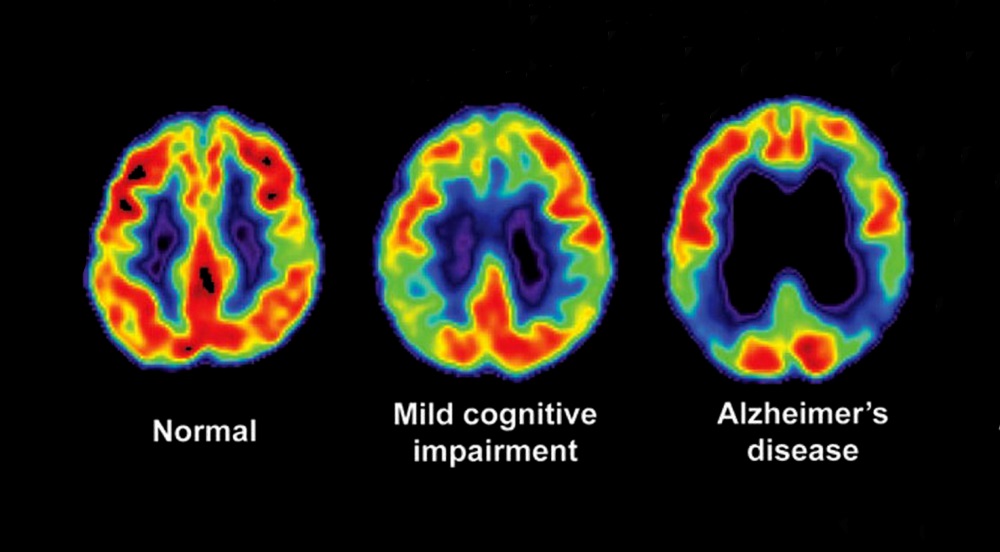
Healthy brain cells take the glucose in for energy production and show up orange or yellow on the scan. Areas involved in the disease cannot take the tracer in and lack the orange/yellow appearance that the glucose tracer creates. The image on the left is an FDG PET scan of a normal brain. The image on the right is of Alzheimer’s disease where the temporal (side) lobes and right frontal lobes are involved. The middle image is of MCI or pre-Alzheimer’s.
Several treatment strategies can be helpful in restoring health energy production in neurons. The first is a ketogenic diet which is the core diet of the Bredesen Protocol. While peripheral cells such as muscle cells can produce energy from fatty acids when glucose is unavailable, neurons cannot. Their only other fuel source is ketones which fats can be converted to.
Another method of restoring energy to neurons is the use of photobiomodulation (PBM). Cells such as neurons have “chromophores” in the energy production pathway. These are enzymes that are sensitive to light which can increase energy production preventing cell death and actually triggering regeneration. The key to brain PBM is using a type of light that can reach into the brain tissue activating the neurons in the involved areas.
The idea that light could be helpful in Alzheimer’s disease first came from studies of animals with large amounts of beta amyloid which is the misfolded protein that generates part of the toxic injury to cells. Lights flashing at 40 Hz caused much of the beta amyloid to be cleared. Unfortunately, this only occurred in the occipital cortex where vision is recorded and interpreted as white light only enters the brain through the optic nerve to the occipital cortex. The temporal and parietal lobes are the brain areas affected in Alzheimer’s disease.
The next phase of research shifted to using infrared light which is a much higher wavelength than white light allowing it to penetrate about 40 mm or 1 ½ inches into the brain where it is applied. Different PBM devices with many diodes have been developed to cover the entire brain including the areas typically affected by the disease. This approach has been shown to be effective in improving cognitive function in both animal studies and human trials.
As would be expected PBM devices have evolved quickly in parallel with this research with more capable devices producing better results. The latest published clinical trial appeared in the July issue of Aging and Disease. In this trial patients with mild to moderate dementia were randomized to receive either active near infrared therapy or a sham procedure for 8 weeks. Prior to beginning the trial subjects had average scores of 23 on the MMSE cognitive test. The test has a maximum of 30 points and scores >25 are normal.
Following the 8 weeks of PBM the control subjects had no significant change in MMSE scores. In contrast subjects receiving the active PBM improved to a mean of 27.6, which is in the normal range. No adverse effects were seen in the trial of 66 subjects receiving active treatment.

This trial used the latest generation transcranial near-infrared PBM device which covers the entire brain with multiple diodes that emit 1070 nm light. This wavelength is uniquely efficient at penetration and tissue activation.
Other outcomes of the trial were that caregivers reported significantly less anxiety and better sleep, both problems that are associated with dementias such as Alzheimer’s disease.
One of the interesting observations with Alzheimer’s disease over the past decade is that no single drug seems to be able to importantly slow or reverse the disease. All progress has come from combinations of nutrition/lifestyle change and neuro-rehabilitative techniques such as PBM.